Topics: health
Boston Levin & The Future of Wearable Technology: An Interview With Udit Agarwal
EPAM Systems acquires healthtech company Odysseus Dara Services to enhance life sciences capabilities
Annova Solutions Expands U.S. Operations with New Colorado Office
Transforming Diagnostics in the EU: The Synergy of IVD and IHD Tests under IVDR
Cloud-powered Healthcare Transforming Patient-centric Solutions
Almirall S.A. Begins First Phase Clinical Study Of ALM223
NCCN and SLACOM Host International Symposium to Improve Breast Cancer Care in Latin America
Experts from across Latin America explore how to improve access to recommended treatment practices based on the latest international guidelines.
BUENOS AIRES, Argentina and PLYMOUTH MEETING, Pa., Nov. 30, 2023 /PRNewswire/ -- Today the Latin American and Caribbean Society of Medical Oncology (SLACOM) and National Comprehensive Cancer Network® (NCCN®) host the Latin American Regional Breast Cancer Summit: Advocating and Implementing Guideline-Concordant Cancer Care for Patients. Esteemed experts from Argentina, Brazil, Mexico, Columbia, Peru, and the United States will present challenges, barriers, and potential solutions for improving access to guideline-concordant breast cancer care in the Latin American region. The regional summit provides an opportunity for a diverse group of multidisciplinary experts to identify and fill any knowledge gaps in breast cancer care and health system processes for cancer prevention and control in Latin America.

Visit NCCN.org/global for cancer treatment resources that have been tailored for specific regions or translated into Spanish, Portuguese, and other languages.
"There is a clear and urgent need to improve our current processes and systems, including those that guide patients in navigating health systems in Latin America," said Eduardo L. Cazap, MD, PhD, Founder and First President of SLACOM. "We hope that the findings and recommendations from the SLACOM/NCCN Working Group, as well as the feedback from the Latin American Regional Breast Cancer Summit will inform the development of Implementation Guidelines for Breast Cancer that can be used by patients, physicians, and systems to take action to address the needs of patients."
SLACOM and NCCN first launched a project to convene thought leaders, government entities, health care providers, patients, and civil society members to conduct a rigorous analysis of cancer burden, existing guidelines and healthcare utilization and accessibility, gaps, obstacles, and system disruptions to guideline-concordant care in Argentina in 2022. The SLACOM/NCCN Working Group developed a set of findings and recommendations to address the major impediments to guideline implementation in breast cancer care, which were presented and discussed at the summit.
That summit also included a presentation outlining the World Health Organization (WHO) Global Breast Cancer Initiative Implementation Framework to provide a practical model to help inform the work of the group moving forward.
"We are honored to continue working alongside SLACOM and others in the Latin America region," said Wui-Jin Koh, MD, Chief Medical Officer, NCCN. "People with cancer everywhere should have access to treatment based on established evidence-based, expert consensus guidelines. Today's webinar is part of the SLACOM/NCCN Working Group's ongoing commitment to develop procedures that improve the implementation of best practices in this region, according to the latest international standards."
NCCN has previously collaborated with other groups in the Latin American region and translated resources for patients and health care providers into Spanish and other languages. Learn more about how the gold standard NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) have been adapted for various locations and resource levels worldwide at NCCN.org/global.
About the National Comprehensive Cancer Network
The National Comprehensive Cancer Network® (NCCN®) is a not-for-profit alliance of leading cancer centers devoted to patient care, research, and education. NCCN is dedicated to improving and facilitating quality, effective, equitable, and accessible cancer care so all patients can live better lives. The NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) provide transparent, evidence-based, expert consensus recommendations for cancer treatment, prevention, and supportive services; they are the recognized standard for clinical direction and policy in cancer management and the most thorough and frequently-updated clinical practice guidelines available in any area of medicine. The NCCN Guidelines for Patients® provide expert cancer treatment information to inform and empower patients and caregivers, through support from the NCCN Foundation®. NCCN also advances continuing education, global initiatives, policy, and research collaboration and publication in oncology. Visit NCCN.org for more information.
Media Contact:
Rachel Darwin
267-622-6624
darwin@nccn.org
Logo - https://mma.prnewswire.com/media/441768/NCCN_Logo.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/nccn-and-slacom-host-international-symposium-to-improve-breast-cancer-care-in-latin-america-301996263.html
View original content:https://www.prnewswire.co.uk/news-releases/nccn-and-slacom-host-international-symposium-to-improve-breast-cancer-care-in-latin-america-301996263.html

AI reality vs. myth: Twelve predictions from SAS for 2024
AI will not take all jobs nor end civilization. But it will help businesses make better decisions.
CARY, N.C., Nov. 30, 2023 /PRNewswire/ -- Artificial intelligence (AI) is everywhere. And stories are rampant about its promise and its threat. Will AI's potential be realized in the year ahead? SAS, the leader in AI and analytics, asked executives and experts across the company to predict trends and key business and technology developments in AI for 2024. Below are some of the predictions they shared.
Visit SAS' 2024 AI predictions page for more trends and forecasts.
Generative AI will augment (not replace) a comprehensive AI strategy
"Generative AI technology does a lot of things, but it can't do everything. In 2024, organizations will pivot from viewing generative AI as a stand-alone technology to integrating it as a complement to industry-specific AI strategies. In banking, simulated data for stress testing and scenario analysis will help predict risks and prevent losses. In health care, that means the generation of individualized treatment plans. In manufacturing, generative AI can simulate production to identify improvements in quality, reliability, maintenance, energy efficiency and yield."
– Bryan Harris, Chief Technology Officer, SAS
[Note: Earlier this year, SAS committed $1 billion to AI-powered industry solutions.]
AI will create jobs
"In 2023, there was a lot of worry about the jobs that AI might eliminate. The conversation in 2024 will focus instead on the jobs AI will create. An obvious example is prompt engineering, which links a model's potential with its real-world application. AI helps workers at all skill levels and roles to be more effective and efficient. And while new AI technologies in 2024 and beyond may cause some short-term disruptions in the job market, they will spark many new jobs and new roles that will help drive economic growth."
– Udo Sglavo, Vice President of Advanced Analytics, SAS
AI will enhance responsible marketing
"As marketers we must consciously practice responsible marketing. Facets of this are awareness of the fallibility of AI and alertness to possible bias creeping in. While AI offers the promise of enhanced marketing and advertising programs, we know that biased data and models beget biased results. In SAS Marketing, we are implementing model cards that are like an ingredient list, but for AI. Whether you create or apply AI, you are responsible for its impact. That's why all marketers, regardless of technical know-how, can review the model cards, validate that their algorithms are effective and fair, and adjust as needed."
– Jennifer Chase, Chief Marketing Officer, SAS
Financial firms will embrace AI amid a Dark Age of Fraud
"Even as consumers signal increased fraud vigilance, generative AI and deepfake technology are helping fraudsters hone their multitrillion-dollar craft. Phishing messages are more polished. Imitation websites look stunningly legitimate. A crook can clone a voice with a few seconds of audio using simple online tools. We are entering the Dark Age of Fraud, where banks and credit unions will scramble to make up for lost time in AI adoption – incentivized, no doubt, by regulatory shifts forcing financial firms to assume greater liability for soaring APP [authorized push payment] scams and other frauds."
– Stu Bradley, Senior Vice President of Risk, Fraud and Compliance Solutions, SAS
Shadow AI will challenge CIOs
"CIOs have struggled with 'shadow IT' in the past and will now confront 'shadow AI' – solutions used by or developed within an organization without official sanction or monitoring by IT. Well-intentioned employees will continue to use generative AI tools to increase productivity. And CIOs will wrestle daily with how much to embrace these generative AI tools and what guardrails should be put in place to safeguard their organizations from associated risks."
– Jay Upchurch, Chief Information Officer, SAS
Multimodal AI and AI simulation will reach new frontiers
"The integration of text, images and audio into a single model is the next frontier of generative AI. Known as multimodal AI, it can process a diverse range of inputs simultaneously, enabling more context-aware applications for effective decision making. An example of this will be the generation of 3D objects, environments and spatial data. This will have applications in augmented reality [AR], virtual reality [VR], and the simulation of complex physical systems such as digital twins."
– Marinela Profi, AI/Generative AI Strategy Advisor, SAS
Digital-twin adoption will accelerate
"Technologies like AI and IoT [Internet of Things] analytics drive important sectors of the economy, including manufacturing, energy and government. Workers on the factory floor and in the executive suite use these technologies to transform huge volumes of data into better, faster decisions. In 2024, the adoption of AI and IoT analytics will accelerate through broader use of digital-twin technologies, which analyze real-time sensor and operational data and create duplicates of complex systems like factories, smart cities and energy grids. With digital twins, organizations can optimize operations, improve product quality, enhance safety, increase reliability and reduce emissions."
– Jason Mann, Vice President of IoT, SAS
Insurers will confront climate risk, aided by AI
"After decades of anticipation, climate change has transformed from speculative menace to genuine threat. Global insured losses from natural disasters surpassed $130 billion in 2022, and insurers worldwide are feeling the squeeze. US insurers, for example, are under scrutiny for raising premiums and withdrawing from hard-hit states like California and Florida, leaving tens of millions of consumers in the lurch. To survive this crisis, insurers will increasingly adopt AI to tap the potential of their immense data stores to shore up liquidity and be competitive. Beyond the gains they realize in dynamic premium pricing and risk assessment, AI will help them automate and enhance claims processing, fraud detection, customer service and more."
– Troy Haines, Senior Vice President of Risk Research and Quantitative Solutions, SAS
AI importance will grow in government
"The workforce implications of AI will start being felt in government. Governments have a hard time attracting and retaining AI talent since experts command such high salaries, however, they will aggressively recruit for expertise to support regulatory actions. And like enterprises, governments will also increasingly turn to AI and analytics to boost productivity, automate menial tasks and mitigate that talent shortage."
– Reggie Townsend, Vice President of the SAS Data Ethics Practice
Generative AI will bolster patient care
"To advance health and improve patient and member experiences, organizations will further develop generative AI-powered tools in 2024 for personalized medicine, such as the creation of patient-specific avatars for use in clinical trials and the generation of individualized treatment plans. Additionally, we will see the emergence of generative AI-based systems for clinical decision support, delivering real-time guidance to payers, providers and pharmaceutical organizations."
– Steve Kearney, Global Medical Director, SAS
Deliberate AI deployment will make or break insurers
"In 2024, one of the top 100 global insurers will go out of business as a consequence of deploying generative AI too quickly. Right now, insurers are rolling out autonomous systems at breakneck speed with no tailoring to their business models. They're hoping that using AI to crunch through claims quickly will offset the last few years of poor business results. But after 2023's layoffs, remaining staff will be spread too thin to enact the necessary oversight to deploy AI ethically and at scale. The myth of AI as a cure-all will trigger tens of thousands of faulty business decisions that will lead to a corporate collapse, which may irreparably damage consumer and regulator trust."
– Franklin Manchester, Global Insurance Strategic Advisor, SAS
Public health will get an AI boost from academia
"Public health is achieving technologic modernization at an unprecedented rate. Whether overdoses or flu surveillance, using data to anticipate public health interventions is essential. Forecasting and modeling are rapidly becoming the cornerstone of public health work, but government needs help. Enter academia. We will see an increase in academic researchers carrying out AI-driven modeling and forecasting on behalf of government. It is clear after COVID-19 that the protection of our population will require exceptional technology and collaboration."
– Dr. Meghan Schaeffer, National Public Health Advisor and Epidemiologist, SAS
Want more?
This spring, you can talk with SAS executives about their predictions and explore the latest in AI and analytics. Join business leaders and analytics experts for SAS Innovate, April 16-19, 2024, in Las Vegas. Sign up today for updates on the conference and early-bird pricing.
About SAS
SAS is a global leader in AI and analytics software, including industry-specific solutions. SAS helps organizations transform data into trusted decisions faster by providing knowledge in the moments that matter. SAS gives you THE POWER TO KNOW®.
SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc. in the USA and other countries. ® indicates USA registration. Other brand and product names are trademarks of their respective companies. Copyright © 2023 SAS Institute Inc. All rights reserved.
Editorial Contact:
Mike Nemecek
SAS
mike.nemecek@sas.com
919-531-5140
sas.com/news


Video - https://mma.prnewswire.com/media/2288686/SAS_AI_predictions_2024_video.mp4
Photo - https://mma.prnewswire.com/media/2288685/SAS_AI_predictions_2024.jpg
Logo - https://mma.prnewswire.com/media/1250367/4429871/SAS_v1_Logo.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/ai-reality-vs-myth-twelve-predictions-from-sas-for-2024-302001272.html
View original content:https://www.prnewswire.co.uk/news-releases/ai-reality-vs-myth-twelve-predictions-from-sas-for-2024-302001272.html

Biocon Biologics Successfully Completes Integration of Viatris’ Biosimilar Business in 31 Countries in Europe
BENGALURU, India, Nov. 30, 2023 /PRNewswire/ -- Biocon Biologics Ltd (BBL), a subsidiary of Biocon Ltd (BSE code: 532523, NSE: BIOCON), today announced that the integration of the Viatris' biosimilars business in 31 European countries has been successfully completed.

Following the acquisition of substantially all of the global biosimilar business of Viatris in November 2022, and the related integration of over 70 emerging market countries in July 2023 and North America in September 2023, the full transition of Viatris' biosimilars operations to Biocon Biologics Ltd in Europe represents another significant milestone as a world-leading biosimilars company.
In Europe, Biocon Biologics portfolio includes 7 biosimilars: Insulin Aspart and Insulin Glargine, Bevacizumab, Pegfilgrastim, Trastuzumab, Adalimumab, and Etanercept. At a global level, Biocon Biologics has a robust pipeline of 20 assets for diabetology, oncology, immunology, ophthalmology, and bone health. Serving over 5.7M patients annually, the Company is committed to providing access to high-quality therapies and solutions to patients, healthcare systems and governments across the globe.
Shreehas Tambe, CEO & Managing Director, Biocon Biologics Ltd, said: "The integration of the Viatris' biosimilar business in Europe ahead of schedule is another important milestone for Biocon Biologics in our journey as a global biosimilars leader. We are pleased to expand access to life-saving treatments for patients across Europe. Our unique, fully integrated capabilities and robust pipeline of 20 products will allows us to better address patient needs and be a reliable partner to health organisations."
Jozef Belcik, Head of Commercial Europe & JANZ, Biocon Biologics Ltd, said: "We are extremely pleased to have reached this historic moment and we are proud to lead the way in the advancement of biosimilars. As we continue to drive innovation and invest in the future, our mission is singularly focused on the development, manufacturing, distribution, and commercialization of biosimilars in Europe and in the world."
After the acquisition from Viatris, Biocon Biologics and its partners will commercialize biosimilar products in the following countries: Albania, Austria, Belgium, Bosnia, Bulgaria, Croatia, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Malta, Netherlands, Norway, Poland, Portugal, Romania, Serbia, Slovakia, Slovenia, Spain, Sweden, Switzerland, and The United Kingdom.
Note about biosimilars:
Based on IQVIA White Paper "The impact of biosimilar competition in Europe, IQVIA (2022)", as of 2022, the cumulative savings at list prices from the impact of biosimilar competition in Europe reached over €30 billion. Furthermore, since the launch of biosimilars these provided nearly 4.5 billion patient treatment days to European patients.
Source:
1-The impact of biosimilar competition in Europe, IQVIA (2022)
December 2022. https://www.iqvia.com/-/media/iqvia/pdfs/library/white-papers/the-impact-of-biosimilar-competition-in-europe-2022.pdf
About Biocon Biologics Limited:
Biocon Biologics Ltd. (BBL), a subsidiary of Biocon Ltd., is a unique, fully integrated, global biosimilars company committed to transforming healthcare and transforming lives by enabling affordable access to high quality biosimilars for millions of patients worldwide. It is leveraging cutting-edge science, innovative tech platforms, global scale manufacturing capabilities and world-class quality systems to lower costs of biological therapeutics while improving healthcare outcomes.
BBL has acquired substantially all of the global biosimilars business of its long-standing partner Viatris, which is a historic milestone in its value creation journey. Biocon Biologics has commercialized eight biosimilars in key emerging markets and advanced markets like U.S., Europe, Australia, Canada, and Japan.
The Company has a pipeline of 20 biosimilar assets across diabetology, oncology, immunology, ophthalmology, and other non-communicable diseases. It has many 'firsts' to its credit in the biosimilars industry. As part of its environmental, social and governance (ESG) commitment, BBL is advancing the health of patients, people, and the planet to achieve key UN Sustainable Development Goals (SDGs). Website: www.bioconbiologics.com; Follow us on LinkedIn: Biocon Biologics ; Twitter: @BioconBiologics for company updates.
Biocon Limited, publicly listed in 2004, (BSE code: 532523, NSE Id: BIOCON, ISIN Id: INE376G01013) is an innovation-led global biopharmaceuticals company committed to enhance affordable access to complex therapies for chronic conditions like diabetes, cancer and autoimmune. It has developed and commercialized novel biologics, biosimilars, and complex small molecule APIs in India and several key global markets as well as Generic Formulations in the US, Europe & key emerging markets. It also has a pipeline of promising novel assets in immunotherapy under development. Website: www.biocon.com; Follow-us on Linked In: Biocon Limited; Twitter: @bioconlimited for company updates.
Forward -Looking Statements: Biocon
This press release may include statements of future expectations and other forward-looking statements based on management's current expectations and beliefs concerning future developments and their potential effects upon Biocon and its subsidiaries/ associates. These forward-looking statements involve known or unknown risks and uncertainties that could cause actual results, performance or events to differ materially from those expressed or implied in such statements. Important factors that could cause actual results to differ materially from our expectations include, amongst other: general economic and business conditions in India and overseas, our ability to successfully implement our strategy, our research and development efforts, our growth and expansion plans and technological changes, changes in the value of the Rupee and other currency changes, changes in the Indian and international interest rates, change in laws and regulations that apply to the Indian and global biotechnology and pharmaceuticals industries, increasing competition in and the conditions of the Indian and global biotechnology and pharmaceuticals industries, changes in political conditions in India and changes in the foreign exchange control regulations in India. Neither Biocon, nor our Directors, or any of our subsidiaries/associates assume any obligation to update any particular forward-looking statement contained in this release.
Logo - https://mma.prnewswire.com/media/2143906/Biocon_Biologics_Logo.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/biocon-biologics-successfully-completes-integration-of-viatris-biosimilar-business-in-31-countries-in-europe-302001922.html
View original content:https://www.prnewswire.co.uk/news-releases/biocon-biologics-successfully-completes-integration-of-viatris-biosimilar-business-in-31-countries-in-europe-302001922.html

Unither Pharmaceuticals celebrates 30 years of innovation and expertise
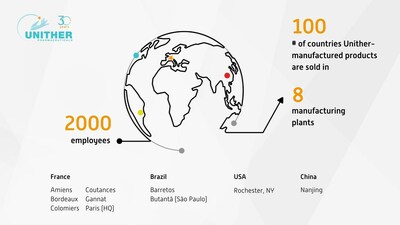
PARIS, Nov. 29, 2023 /PRNewswire/ -- Unither Pharmaceuticals, a leading CDMO (Contract Development and Manufacturing Organization), celebrates 30 years in business. In 1993, Unither Pharmaceuticals began operations by acquiring a 17-man pharmaceutical plant that produced 3 pharmaceutical forms. Today, the company is a world leader in the manufacture of sterile unit doses, with a production capacity of 5 billion doses and a presence on four continents. This growth is also reflected in rising sales, which should reach 500 million euros by 2023.
To view the Multimedia News Release, please click:
https://www.multivu.com/players/uk/9229051-unither-pharmaceuticals-celebrates-30-years-innovation-expertise/
Unither first implemented Blow-Fill-Seal (BFS) technology in 1995, enabling them to manufacture sterile doses of saline solutions, asthma medications, and eye drops. From 1997 onwards, the company developed its international business, first by exporting its products, then by acquiring a factory in the United States in 2013. With the acquisition of Barretos in 2015, Unither gained a foothold in Brazil. Then in 2020, an acquisition in China, inNanjing, consolidated its global footprint. In 2022, Unither further expanded its activities with the acquisition of a new site specializing in ophthalmology in São Paulo. This international development is designed to support its customers' international industrial strategies by getting closer to their target markets.
Since its inception, Unither has been at the forefront of innovation, continuously striving to improve patients' lives. Eric Goupil, President of Unither Pharmaceuticals, reminds the group's vision: "We want to be recognized for making healthcare solutions that improve and simplify patients' lives accessible to as many people as possible. We imagine new ideas, and our enthusiasm and energy enable us to realize them for the benefit of our customers.".
Jean-François Hilaire, CEO of Unither Pharmaceuticals, looks to the future: "Unither's vocation is to support our customers in their drive for innovation and in their efforts to meet the demands of their markets. With Euroject®, our recent breakthrough in merging a single-use dose with a needle to create a ready-to-use injection system, illustrates Unither's commitment to simplifying the lives of healthcare practitioners and patients, while keeping costs under control.".
About Unither Pharmaceuticals: www.unither-pharma.com/about/who-are-we/
Press contact:
contact@unither-pharma.com

Video - https://www.youtube.com/watch?v=lNCXXx_8wTo
Photo - https://mma.prnewswire.com/media/2287080/Unither_Pharmaceuticals.jpg
Logo - https://mma.prnewswire.com/media/1471719/Unither_Pharmaceuticals_Logo.jpg
![]() View original content:https://www.prnewswire.co.uk/news-releases/unither-pharmaceuticals-celebrates-30-years-of-innovation-and-expertise-302001064.html
View original content:https://www.prnewswire.co.uk/news-releases/unither-pharmaceuticals-celebrates-30-years-of-innovation-and-expertise-302001064.html



